Search results
Search for "heterocyclic amino acids" in Full Text gives 2 result(s) in Beilstein Journal of Organic Chemistry.
Regioselective synthesis of methyl 5-(N-Boc-cycloaminyl)-1,2-oxazole-4-carboxylates as new amino acid-like building blocks
Beilstein J. Org. Chem. 2022, 18, 102–109, doi:10.3762/bjoc.18.11
- analysis, 1H, 13C, and 15N NMR spectroscopy, HRMS, and single-crystal X-ray diffraction data. Keywords: β-enamino ketoesters; heterocyclic amino acids; 15N-labeled 1,2-oxazole; NMR (1H; 13C; 15N); 1,2-oxazole (isoxazole); X-ray structure analysis; Introduction 1,2-Oxazoles (isoxazoles) constitute an
- peptide-like structure. Heterocyclic amino acids and related compounds have been used to prepare synthetic DNA-encoded compound libraries for the discovery of small molecule protein ligands [23][24][25]. Recently, a highly specific and potent p38α kinase inhibitor containing a 3-amino-1-phenyl-1H-pyrazole
Photocontrolled DNA minor groove interactions of imidazole/pyrrole polyamides
Beilstein J. Org. Chem. 2020, 16, 60–70, doi:10.3762/bjoc.16.8
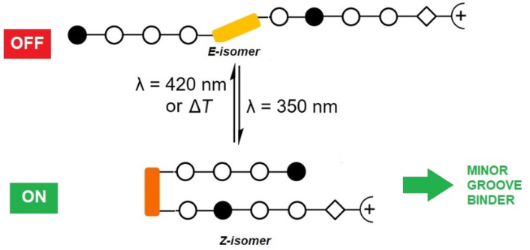
- systematically increased the number of binding motifs, and thus achieved sequence-specific binding [6][7][8][9]. Selectively binding polyamides adopt an antiparallel hairpin structure where a base pair of the DNA is addressed by a pair of the heterocyclic amino acids in the hairpin assembly. Overall, specific
- required because this allowed for an alignment between hydrogen-bonding groups in long polyamides and in the minor groove of DNA [41]. The Fmoc-protected heterocyclic amino acids 2 were obtained from N-methylpyrrole and N-methylimidazole, respectively (Scheme 2A). The N-terminal N-methylpyrrole and N























